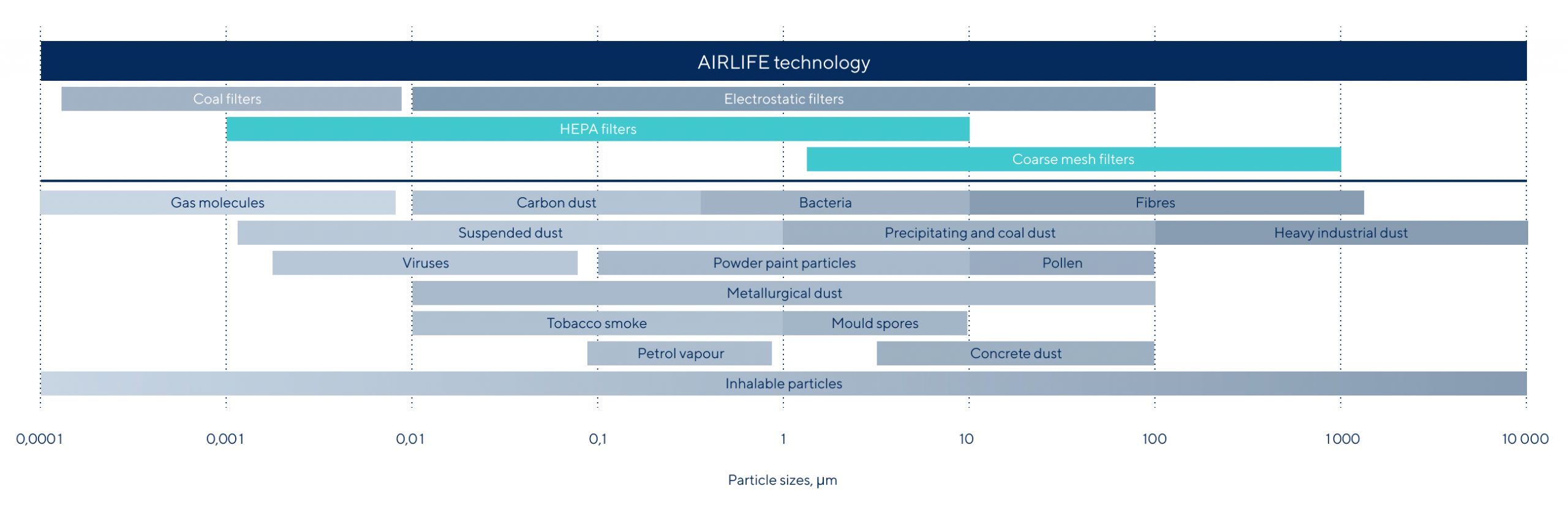
Technology
Our unique patented technology for air purification and disinfection – the foundation of all AL air purifiers – is the result of a research collaboration with some of the world’s leading universities.

Approved by independent laboratories and used daily in all types of environments, this AL technology purifies the air of all types of pollutants – mechanical (aerosols), microbiological, and chemical.
-
Pre-filter– This filter retains large particles of dust and ash. Filtration class M5-M6 (EN 1822, EN 799).
-
Aerosol charging unit– The unit ensures a symmetrical electrostatic field inside the element and charges 99.8% of aerosols with sizes between 0.1-100 μm and a linear velocity of up to 4 m/s.
-
Combined polarized photocatalytic filter– The filter traps particles up to 0.1 μm in size, adsorbs unpleasant odors, toxic chemical substances, allergens, viruses, and bacteria. The polarized filter causes the charged particles to precipitate, reaching a filtration efficiency of 99.9% with minimal airflow resistance. The photocatalyst uses soft UV radiation to oxidize organic compounds into harmless components – carbon dioxide, water, and atmospheric nitrogen.
-
UV-A emitters– UV radiation provides the energy for activating the photocatalyst. The device’s UV lamps emit UV light with wavelengths in the range of 320-400 nm (spectrum A).
-
Combined adsorption catalytic filter– This filter prevents penetration of harmful substances by adsorbing them on the surface of the catalytically active, modified carbon sorbent. The joint action of the adsorption and photocatalytic filtration stages makes the adsorbent catalytically effective, and it regenerates during operation.
Air treatment efficiency
(dust, aerosols, allergens, soot, radioactive aerosols, combustion products)
| Particle size (μm) | Efficiency % |
|---|---|
| > 0,3 μm | 99,87 |
| > 0,5 μm | 99,96 |
| > 0,7 μm | 99,99 |
| > 1,0 μm | 99,99 |
| > 3,0 μm | 99,999 |
| > 5,0 μm | 99,999 |
| 10,0 μm | 99,99999 |
The tables show the one-pass air purification efficiency of the AL SA-100 filtration system.
| Title | Type | Efficiency % |
|---|---|---|
| Staphylococcus aureus | Bacterium | 99.999 |
| Bacillus anthracis | Bacterium | 99.940 |
| Staphylococcus epidermidis | Bacterium | 99.990 |
| H1/N1 | Virus | 99.999 |
| Poliovirus 1 | Virus | 99.999 |
| Stachybotrys chartarum | Fungus spores | 99.960 |
| Aspergillus fumigatus | Fungus spores | 99.870 |
| Aspergillus niger | Fungus spores | 99.800 |
| Mycobacterium tuberculosis | Mycobacterium | 99.990 |
| Coronavirus | Virus | 99.999 |
The tables show the one-pass air purification efficiency of the AL SA-100 filtration system.
| Substance | Input concentration, mg/m³ |
Output concentration, mg/m³ |
One-pass efficiency, % |
| Carbon monoxide (СО) | 25.14 | 1.030 | 95.90 |
| Ammonia (NH3) | 250.00 | 0.190 | 99.92 |
| Formaldehyde (СН2О) | 9.00 | 0.005 | 99.94 |
| Ozone (О3) | 35.50 | 0.004 | 99.99 |
| Benzol (С6Н6) | 42.55 | 0.070 | 99.84 |
| Toluene (C6H5-CH3) | 23.40 | 0.010 | 99.96 |
| Styrene (C8H8) | 11.75 | 0.001 | 99.99 |
| Nitrogen dioxide (NO2) | 74.10 | 0.040 | 99.95 |
| Acetone (C3H6O) | 95.55 | 0.020 | 99.98 |
| Hydrogen sulfide (HS) | 0.012 | 0.0003 | 97.50 |
The tables show the one-pass air purification efficiency of the AL SA-100 filtration system.
| Aspect | UV-C | AL |
| Microorganism mutation | ✘ Possible | ✓ Impossible |
| Odor removal | ✘ Impossible | ✓ Efficient |
| Purification of non-organic compounds | ✘ Impossible | ✓ Efficient |
| Selectivity | ✘ Different radiation doses for different microorganisms | ✓ Oxidation of any organic matter |
| Ozone generation | ✘ Possible, if a lamp with a maximum wavelength of 195 nm is used | ✓ Ozone-free |
| Residual macromolecules | ✘ Organic residual from microorganisms | ✓ Complete oxidation to the simplest compounds |
| Operation mode | ✘ No more than 8 h/day | ✓ 24/7 |
| Aspects | НЕРА | AL |
| Microorganism inactivation | ✘ Impossible | ✓ Efficient |
| Destruction of odor and allergens | ✘ Impossible | ✓ Efficient |
| Destruction of non-organic contaminants | ✘ Impossible | ✓ Efficient |
| Filter capacity | ✘ Low | ✓ High |
| Airflow resistance | ✘ High | ✓ Low |
Photocatalysis
All AL air purification and disinfection systems use photocatalysis to oxidize and inactivate the chemical and microbiological pollutants in air.
Today photocatalysis is defined as “acceleration or initiation of chemical reactions under exposure to light in the presence of photocatalysts that, as a result of adsorbing quantum of light, are capable of causing chemical transformation of the reagents, forming interim chemical compounds with the latter, and regenerating their chemical composition after each such interaction cycle”.
The concept of the photocatalytic purification method is the oxidation of substances on the surface of a catalyst under soft spectrum A UV-radiation (320-400 nm). As purification reactions occur at room temperature, toxic impurities do not accumulate on the purifier filter but decompose to harmless air components, such as carbon dioxide, water, and atmospheric nitrogen.
Photocatalysis principle
Harmful organic, non-organic, and trace element contaminants, as well as bacteria, viruses, and fungi spores get adsorbed on the surface of a photocatalyst applied to a porous foundation (photocatalytic filter). Exposed to the light range emitted by the UV lamp (320-400 nm), they oxidize to carbon dioxide, water, and atmospheric nitrogen.
Used as a catalyst, modified titanium dioxide (TiO2) is a semiconductor compound. In such compounds, electrons are believed to exist both in free and bound states. In the first case, the electrons move on the lattice formed by Ti4+ cations and O2- oxygen anions. In the latter case, the electrons are mostly bound with the lattice ion and form part of a chemical bond. Freeing such an electron requires at least 3.2 eV of energy. Such energy may be supplied by a light quantum with a wavelength of 320-400 nm.
This means that, as light is absorbed by a TiO2 particle, a free electron and an electron vacancy are formed. In semiconductor physics, such a vacancy is called an electron hole.
Both the electron and the hole are quite mobile. Moving within the semiconductor particle, part of them recombines, but part jumps to the surface and gets trapped by it.
The electron and the hole trapped by the surface are quite definite chemical particles. For instance, the electron may be Ti3+ on the surface, while the hole is localized on the lattice oxygen, making O-. As a result, some extremely chemically reactive particles appear on the oxide surface. In terms of oxidation-reduction potential, the reactivity of the electron and the hole on the TiO2 surface is characterized with the following values: the electron potential is ~ -0.1 V, and the hole potential is ~ +3 V relative to a standard hydrogen electrode.